
So, potassium has the largest atomic radii. “Atomic Screening Constants from SCF Functions. The detonation was astronomically powerfulover 1,570 times more powerful, in fact, than the combined two bombs dropped on Hiroshima and Nagasaki. Atomic radius increases from top to bottom and decreases from left to right in periodic table. These values were taken from these sources: Empirically measuring the radius of a single atom is a physically difficult task to accomplish and values vary from source to source. This table was optimized to fit on a single sheet of standard 8½x11 piece of paper.Ĭheck out Periodic Table Trends for more information about other trends that appear in the periodic table.Ītomic radii used for this table were calculated values. For best results, choose Landscape as your printing option. 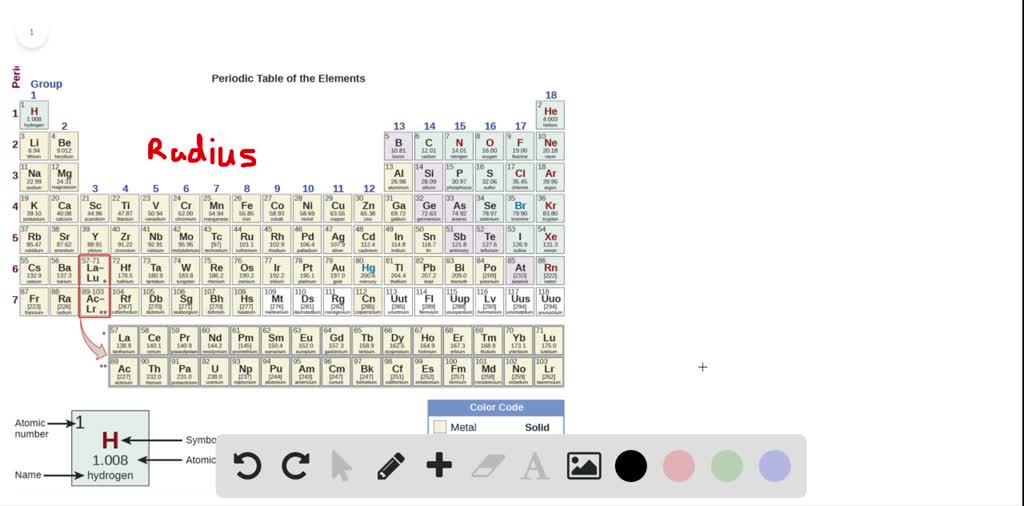
This reduces the force of attraction between the outer electrons and the nucleus and the electron orbits are larger.Ī PDF version of this table is available for printing. These filled shells cancel out part of the positive charge of the nucleus. As you move down the period, more electron shells are filled. This time, even though the number of protons increases by a lot, the electron valence shells do not. Because of these two trends, the largest atoms are found in the lower left corner of the periodic table, and the smallest are found in the upper right corner (Figure 2.8.4). The elements with the largest atomic radii are located towards the left and bottom of the periodic table (i.e. In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. Again, this is due to the effective charge at the nucleus. Answer (1 of 15): You probably meant here which elements are the largest atoms, meaning which elements have the largest atomic radius. This direction increases the size of the atom. The other trend of atomic radius or atom size occurs as you move vertically down an element group. The more pull, the tighter the electrons orbit. The larger charge means a greater pull due to the electrostatic attraction between the positive nucleus and the negative electrons. This increases the overall charge attracting the electrons towards the nucleus. Each step you move to the right on the table, the number of protons in the center increases by one. As you move down the periodic table atoms get bigger. This is due to the way electrons form shells around the nucleus. Which atom has the largest atomic radius potassium Question 2 Q. 
In general, atomic radius or atom size decreases as you move from left to right. In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. This table shows how the atom size, and atomic radius values change as you move horizontally and vertically across the periodic table. So this is the trend for atomic radius.Each atom’s size is relative to the largest element, cesium.Ītom size values are calculated from atomic radius data. 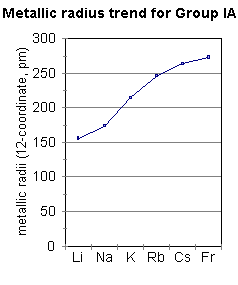
Our largest atomic radius is found in the bottom left corner and our smallest atomic radius is found in the upper right corner. So therefore if it decreases going across and increases going down. With each step we go down each as we go down each step there's an additional energy level added. It increases because we are adding an additional energy level. The largest measured atom is cesium, which has a radius of about 298 picometers. Thus, the correct trend can be shown by option (A) Li < Na < K < Rb < Cs. The atomic radius of an iron atom is about 156 picometers. This shows us that lithium has the smallest atomic radius and caesium has the largest atomic radius within the given elements of a group. The atomic radius of the hydrogen atom is about 53 picometers. So it decreases moving across the period going down a group. How Big Are Atoms A picometer is 1-trillionth of a meter. Therefore pulling the outermost electron are electrons in closer and closer and decreasing that size of the atom. So the nucleus becomes greater and greater in its positive charge. And yet with each step across we add another proton in the nucleus. This is because we are not adding any additional energy levels. So using the periodic table, we need to think about the trend for atomic radius moving across a period. This question asks us to use only a periodic table to determine which of these three elements has the largest atomic ray B.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
